|
On 15 December 2020, the Commission decided to purchase these 80 million additional doses. The Advance Purchase Agreement with Moderna provides for the initial purchase of 80 million doses on behalf of all EU Member States, plus an option to request up to a further 80 million doses. 
This followed a positive scientific recommendation by the European Medicines Agency. On 6 January 2021, the European Commission granted a conditional marketing authorisation for the vaccine developed by Moderna, making it the second vaccine authorised for use in the EU. With the agreement of BioNTech-Pfizer, the redacted Advance Purchase Agreement is available. This agreement ensures that Member States have access to the vaccines when they need them – including future variant-adapted vaccines if authorised, so that they can quickly respond to any epidemiological developments and continue to supporting partner countries globally. On, the European Commission and BioNTech-Pfizer reached an agreement to better address Member States’ needs for COVID-19 vaccines. This will bring the total number of deliveries by BioNTech-Pfizer to 650 million doses in 2022. These doses come on top of the 450 million doses already planned to be delivered in 2022 based on the agreement already signed. The Commission and Member States have also activated a first option to order over 200 million doses under the third contract with BioNTech-Pfizer. :no_upscale()/cdn.vox-cdn.com/uploads/chorus_asset/file/22526360/GettyImages_1278761871.jpg)
On 19 December 2021, the Commission agreed with BioNTech-Pfizer to accelerate deliveries of 20 million vaccine doses in Member States during the first quarter of 2022 (5 million in January, 5 million in February and 10 million in March), bringing the total number of deliveries for this period to 215 million. It will allow for the purchase of 900 million doses of the current vaccine and of a vaccine adapted to variants (if necessary and if approved), with the option to purchase an additional 900 million doses. It reserves an additional 1.8 billion doses on behalf of all EU Member States, between the end of 20. On, the European Commission signed a third contract with BioNTech-Pfizer. On 10 March 2021, the European Commission reached an agreement with BioNTech-Pfizer for the supply of 4 million additional doses of COVID-19 vaccines to tackle coronavirus hotspots and to facilitate free movement across borders. On 8 January 2021, the Commission proposed to Member States to purchase an additional 200 million BioNTech-Pfizer vaccine doses, with the option to acquire another 100 million doses. On 15 December 2020, the Commission decided to purchase these 100 million additional doses. The Advance Purchase Agreement with BioNTech-Pfizer provided for the initial purchase of 200 million doses on behalf of all EU Member States, plus an option to purchase up to a further 100 million doses. On 21 December 2020, the Commission granted a conditional marketing authorisation for the COVID‑19 vaccine developed by BioNTech and Pfizer, making it the first COVID-19 vaccine authorised in the EU. Safe and effective vaccines started to be distributed in the EU by the end of 2020, following strict authorisation procedures and the highest safety standards. 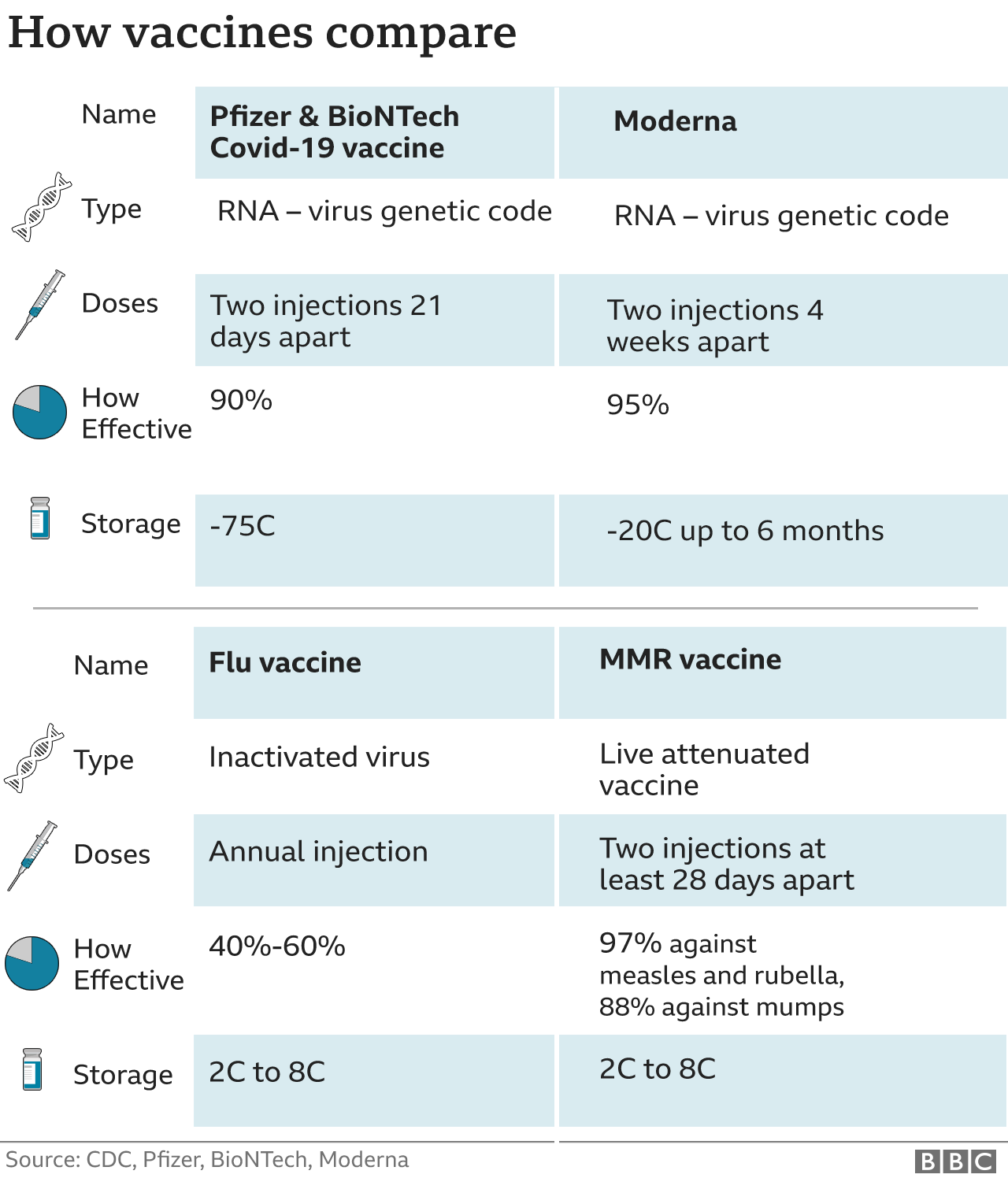
The vaccine strategy should act as a reference point for Member States when formulating their national vaccination strategies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
